Vaccine

Delta Variant: Does the Role of Vitamins Help With Protection Against This COVID-19 Strain?

What Are Booster Shots? US FDA Set to Discuss Pfizer's Application for a COVID-19 Vaccine Third Dose

Pfizer, BioNTech Presents New COVID-19 Vaccines Specialized for Children 5 Years and Up; Seeks Approval for Treatment Use in Age Group
Indian Drug Regulator Approves Use of the ZyCoV-D Vaccine Against COVID-19; The First Authorized DNA-Based Inoculation
Novel Virus-Like Nanoparticle Vaccine Offers New Approach of Protection Against COVID-19
Plant and Bacteria Nanoparticles Used to Develop Fridge-Free COVID-19 Vaccine; Here's How It Works
Ivermectin Not Proven Safe for COVID-19 Treatment; Effects on People's Health Include Overdose, Coma
Influenza Flu Vaccine Recommended for People Aged 6 Months and Older To Avoid ‘Twindemic’
Japan Suspends Over 1.6 Million Moderna Vaccines Due to Particulate Matter Contamination
COVID-19 Delta Variant Spreads Fast; More Travelers From Abroad Want Another Vaccine Booster Shot in the US
BioNTech Plans to Develop New COVID-19 Vaccine Specialized Against the SARS-CoV-2 Delta Variant in Just 4 Weeks or Less
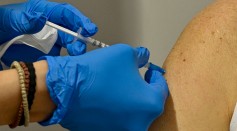
FDA Will Soon Allow 3rd COVID-19 Vaccine Shot for Immunocompromised Persons

CDC Urges Pregnant Women To Get Vaccinated Against COVID-19
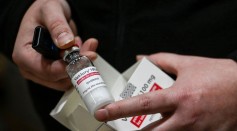
Remdesivir Currently Being Modified from Intravenous to Oral Administration; Study Describes Potential of the Antiviral Drug in Treating COVID-19
Most Popular

Pharaoh's Curse Unlocked? Scientists Claim To Have Cracked the Mystery of What Killed Over 20 People Who Opened the Tomb of King Tut in 1922
Earth's Magnetic Field Nearly Collapsed 600 Million Years Ago, Sparking Evolution of More Complex Life

Why Do People Sigh? Here's What These Deep Breaths Do to the Body and What They Could Mean

Cosmic Monsters Create Intersections That Lead To Stellar-Mass Black Holes Collisions

!['Cosmic Glitch' in Einstein's Theory of General Relativity Could Be Explained in This New Scientific Tweak [Study]](https://1721181113.rsc.cdn77.org/data/thumbs/full/53435/258/146/50/40/cosmic-glitch-in-einsteins-theory-of-general-relativity-could-be-explained-in-this-new-scientific-tweak-study.jpeg)



